FEATURED WORK

Rewiring STAT signaling from the cell surface with Trikine immunotherapeutics
Science (2026)
Abstract: Cytokines dimerize two receptor chains to activate Janus kinases and STAT transcription factors that regulate immune cells but have therapeutic liabilities. We engineered “Trikines” to compel cis formation of three-chain cytokine receptor complexes at the cell surface that induce bespoke STAT transcriptional signaling programs optimized for therapeutic efficacy. Designed Trikines co-activated pSTAT5 and pSTAT3 signatures distinct from any natural cytokines, by assembling trimeric combinations of Interleukin-2 (IL-2), Interleukin-10 (IL-10), and Interleukin-21 (IL-21) receptors. An IL-2-based-Trikine restrained terminal differentiation of T cells, promoted stemness, and enhanced durability of tumor control without toxicity. Unexpectedly, an IL-10-based Trikine induced immune infiltration into poorly immunogenic tumors, showing striking efficacy in small cell lung cancer and pancreatic cancer models. Trikines obviate the need for cell engineering to customize STAT signatures for immunotherapy.

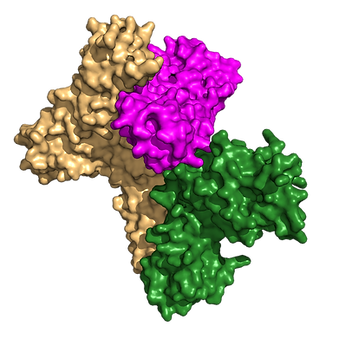
Structure of the interleukin-5 receptor complex exemplifies the organizing principle of common beta cytokine signaling
Molecular Cell (2024)
Abstract: Cytokines regulate immune responses by binding to cell surface receptors, including the common subunit beta (βc), which mediates signaling for GM-CSF, IL-3, and IL-5. Despite known roles in inflammation, the structural basis of IL-5 receptor activation remains unclear. We present the cryo-EM structure of the human IL-5 ternary receptor complex, revealing architectural principles for IL-5, GM-CSF, and IL-3. In mammalian cell culture, single-molecule imaging confirms hexameric IL-5 complex formation on cell surfaces. Engineered chimeric receptors show that IL-5 signaling, as well as IL-3 and GM-CSF, can occur through receptor heterodimerization, obviating the need for higher-order assemblies of βc dimers. These findings provide insights into IL-5 and βc receptor family signaling mechanisms, aiding in the development of therapies for diseases involving deranged βc signaling.